Restylane Skinboosters Vital Light side effects are a key topic for aesthetic doctors and dermatology professionals seeking to enhance skin hydration and elasticity in superficial layers. Manufactured by Galderma, this stabilized hyaluronic acid-based injectable is widely used in facial and neck rejuvenation protocols, with an excellent safety and efficacy profile when applied correctly. This article outlines the product’s clinical profile, expected reactions, injection techniques, patient selection, aftercare, and safety considerations, based on peer-reviewed data and professional guidelines.
What Is Restylane Skinboosters Vital Light?
Restylane Skinboosters Vital Light is a stabilized, non-volumizing hyaluronic acid (HA) injectable, specifically designed for:
- Improving skin quality (hydration, elasticity, smoothness)
- Treating photoaged, thin, or sensitive skin
- Superficial dermal rejuvenation in the face, neck, décolleté, and hands
Product specs:
- HA concentration: 12 mg/ml
- Lidocaine: 3 mg/ml for enhanced comfort
- Volume: 1 x 1 ml prefilled syringe
- Manufacturer: Galderma (Sweden)
- CE-marked Class III medical device
Official site: https://www.restylane.com
How Restylane Skinboosters Work
Unlike volumizing dermal fillers, Skinboosters are injected superficially to hydrate the skin from within. The low HA concentration improves the skin’s water-binding capacity and supports collagen formation over time. With regular sessions, patients often report smoother texture, improved tone, and reduced appearance of fine lines.
Mechanism of action:
- Hyaluronic acid attracts water molecules
- Stimulates fibroblast activity and collagen synthesis
- Gradual dermal remodeling with minimal invasiveness
Clinical studies show that 3 initial sessions followed by maintenance every 6 months yield sustained skin improvements.
Indications and Treatment Areas
| Area Treated | Technique | Depth | Frequency |
|---|---|---|---|
| Face (cheeks) | Micro-puncture or microbolus | Superficial to mid-dermis | 3 sessions every 4 weeks |
| Neck | Microdroplet or linear | Superficial dermis | Maintenance every 6 months |
| Décolleté | Serial puncture | Superficial dermis | Up to 3 sessions |
| Hands | Microbolus | Superficial dermis | As needed, low volume |
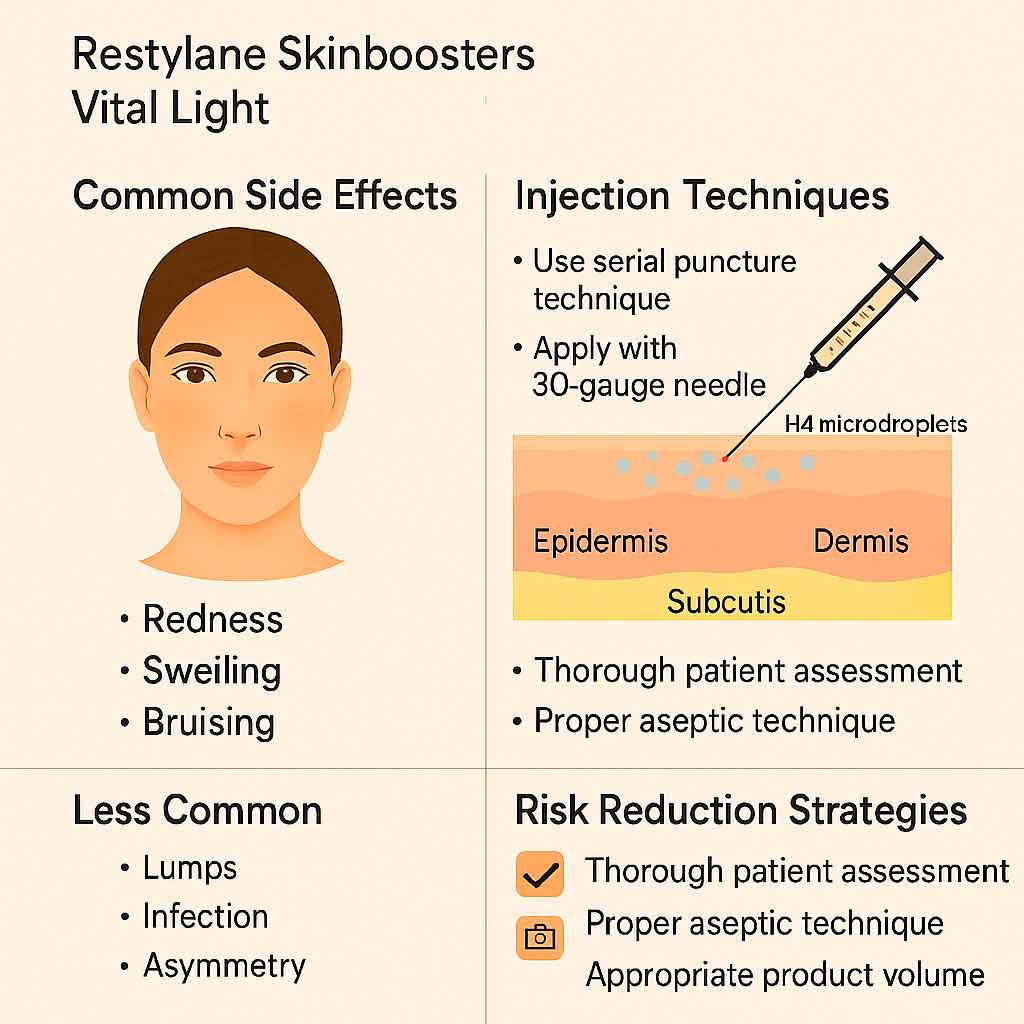
Common and Rare Side Effects of Restylane Skinboosters Vital Light
Table 1: Side Effects Overview
| Side Effect | Frequency | Duration | Notes |
| Redness, swelling | Very common | <48 hours | Expected; mild |
| Bruising | Common | <5 days | Related to technique and depth |
| Tenderness, itching | Occasional | Self-limiting | Minor local irritation |
| Bumps or papules | Uncommon | Temporary | Usually resolves without treatment |
| Delayed inflammation | Rare | Days to weeks | May require corticosteroids |
| Infection or abscess | Very rare | Delayed | Requires antibiotic management |
Sources: Restylane Clinical Manual, PubMed Review on HA Microinjections
Injection Protocol and Best Practices
Recommended needle/cannula: 30G × ½ inch or finer
Injection depth: Superficial to mid dermis
Technique options:
- Serial microinjections
- Microbolus
- Linear threading in fine lines
Tips for safe use:
- Clean injection field thoroughly
- Use low volume per point to avoid lumping
- Instruct patients on post-care hygiene
Avoiding complications:
- Use aspiration in sensitive areas
- Monitor injection depth to avoid vascular events
- Choose candidates without active skin conditions or contraindications
Comparison: Vital Light vs. Standard Skinboosters
| Attribute | Vital Light | Standard Vital |
| HA Concentration | 12 mg/ml | 20 mg/ml |
| Indications | Thin/sensitive skin | Mature/photoaged skin |
| Viscosity | Lower | Higher |
| Comfort | Lidocaine 3 mg/ml included | Optional lidocaine |
| Depth of injection | More superficial | Slightly deeper |
| Use frequency | More frequent for maintenance | Fewer sessions needed |
Ideal Patient Profile and Contraindications
Best suited for:
- Fitzpatrick skin types I–IV
- Fine lines, dehydration, lack of glow
- Younger patients or early aging signs
Contraindications:
- Autoimmune disorders
- Active infection at injection site
- Pregnancy or breastfeeding
- Known hypersensitivity to HA or lidocaine
Patient Education and Informed Consent
It is important to inform patients about:
- Expected mild side effects (e.g., redness, swelling)
- Rare but possible complications (e.g., delayed inflammation)
- Realistic outcomes: Skinboosters improve texture and hydration, not volume
Obtain written informed consent and maintain documentation of treatment plans and batch numbers.
Aftercare Recommendations
- Avoid makeup for 12 hours post-injection
- No heavy exercise or sauna for 48 hours
- Avoid sun exposure for 1 week
- Hydrate adequately and use SPF daily
- Avoid facial massages for 48–72 hours
Summary Table: Product Profile
| Attribute | Detail |
| Product name | Restylane Skinboosters Vital Light |
| HA concentration | 12 mg/ml |
| Lidocaine included | Yes (3 mg/ml) |
| Manufacturer | Galderma |
| Certified use | CE-marked, Class III |
| Injection depth | Superficial to mid dermis |
| Duration of effect | 4–6 months with 3-session protocol |
| Indicated areas | Face, neck, décolleté, hands |
| Treatment purpose | Hydration, elasticity, skin texture |